|
molecular geometry changes to seesaw (four. The VSEPR model assumes that electron pairs in the valence. If asked for the electron-pair geometry on the central atom we must respond with the electron-pair geometry. A table with four rows and six columns is shown.\): The Difference in the Space Occupied by a Lone Pair of Electrons and by a Bonding PairĪs with SO 2, this composite model of electron distribution and negative electrostatic potential in ammonia shows that a lone pair of electrons occupies a larger region of space around the nitrogen atom than does a bonding pair of electrons that is shared with a hydrogen atom. Valence shell electron-pair repulsion theory (VSEPR theory) enables us to predict the molecular structure, including approximate bond angles around a central atom, of a molecule from an examination of the number of bonds and lone electron pairs in its Lewis structure. 2 that the arrangement that minimizes repulsions places the groups 180° apart. atoms are all in the same plane and have a square planar molecular. There are two electron groups around the central atom. Predicting Electron-pair Geometry and Molecular Structure: Lone Pairs on the Central Atom. The sum of these then gives the SN value for the central atom. The central atom, beryllium, contributes two valence electrons, and each hydrogen atom contributes one. In SF 6, each of the 6 valence electrons in sulfur expands to form a bond with a F-atom. In this case, the EPG is described by a octahedron and EPG is called Octahedral. Some expanded valence compounds will have a total of 6 electron pairs (bp & lp) around a central atom. A molecule with the formula AB4 and a tetrahedral molecular geometry uses to form its ô bonds. If asked for the electron-pair geometry on the central atom we must respond with the electron-pair geometry. The molecular geometry is the shape of the molecule. 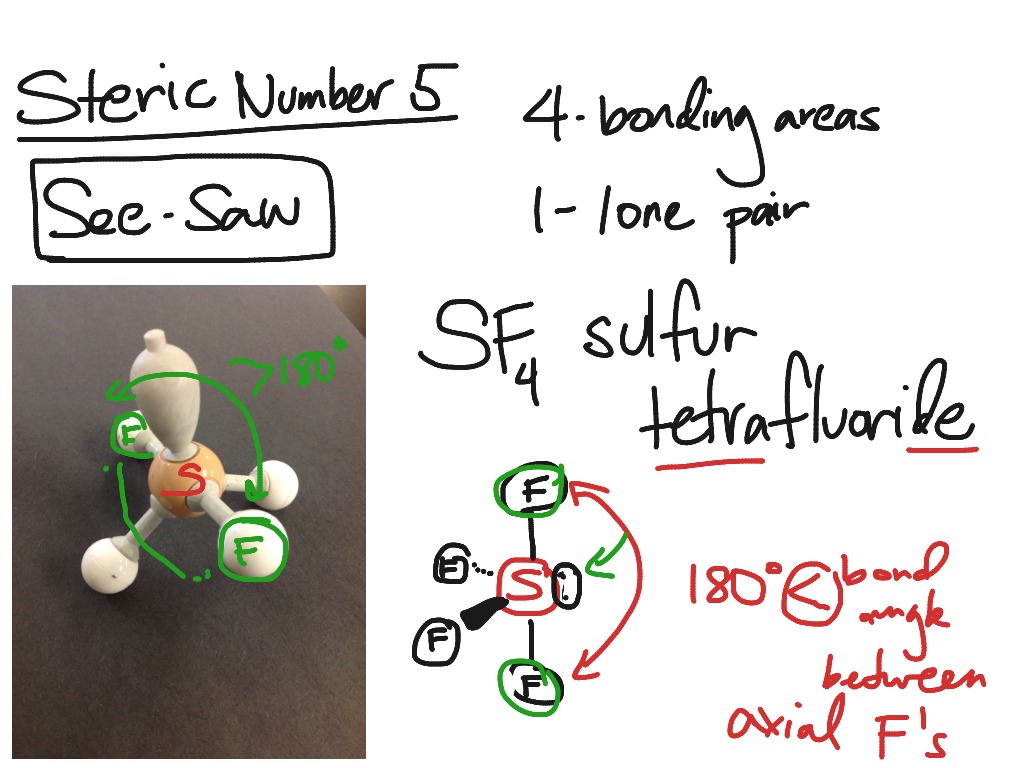 
The molecular geometry of COCl is trigonal planar. The electron-pair geometry provides a guide to the bond angles of between a terminal-central-terminal atom in a compound. The hybridization of central atom ‘C’ is sp². 
Since there are four bond pairs and one lone pair, the molecular geometry of ClF is a see-saw. 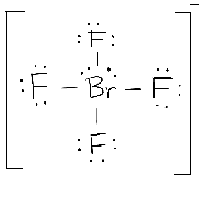
\): The basic electron-pair geometries predicted by VSEPR theory maximize the space around any region of electron density (bonds or lone pairs). The procedure for predicting molecular geometry from a molecular formula consisits of first drawing a valid Lewis structure for the molecule, from which a count of the nonbonding and bonding electron groups (also called electron domains) is obtained. molecular geometry (MG) with both I-I bonds in the axial positions. According to the VSEPR model, a molecule with the general formula AB2 with two lone pairs on the central atom will have a molecular geometry. The electronic geometry of ClF is trigonal bipyramidal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
